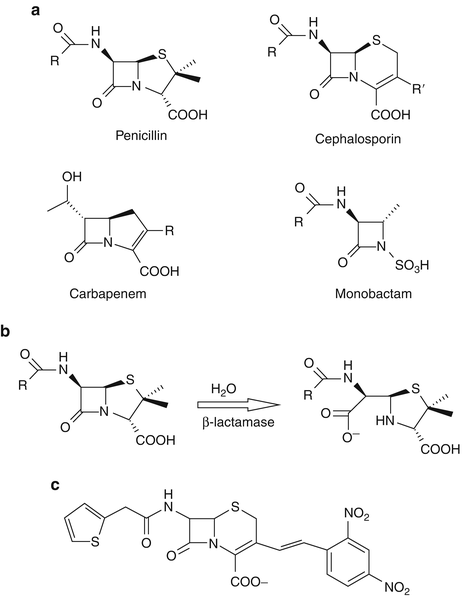
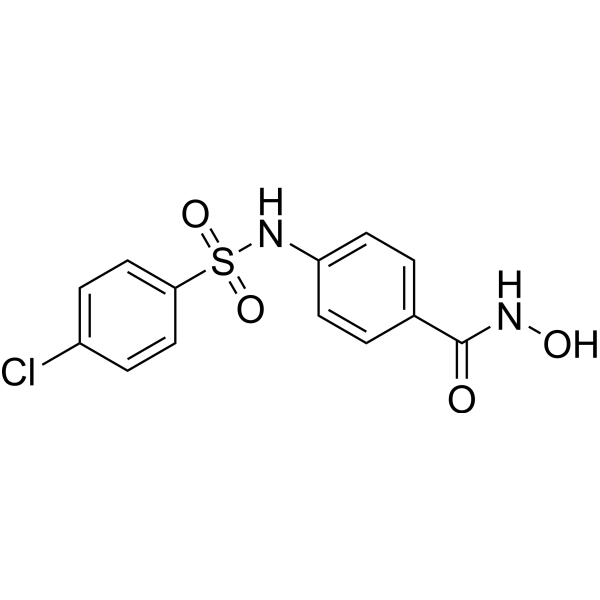
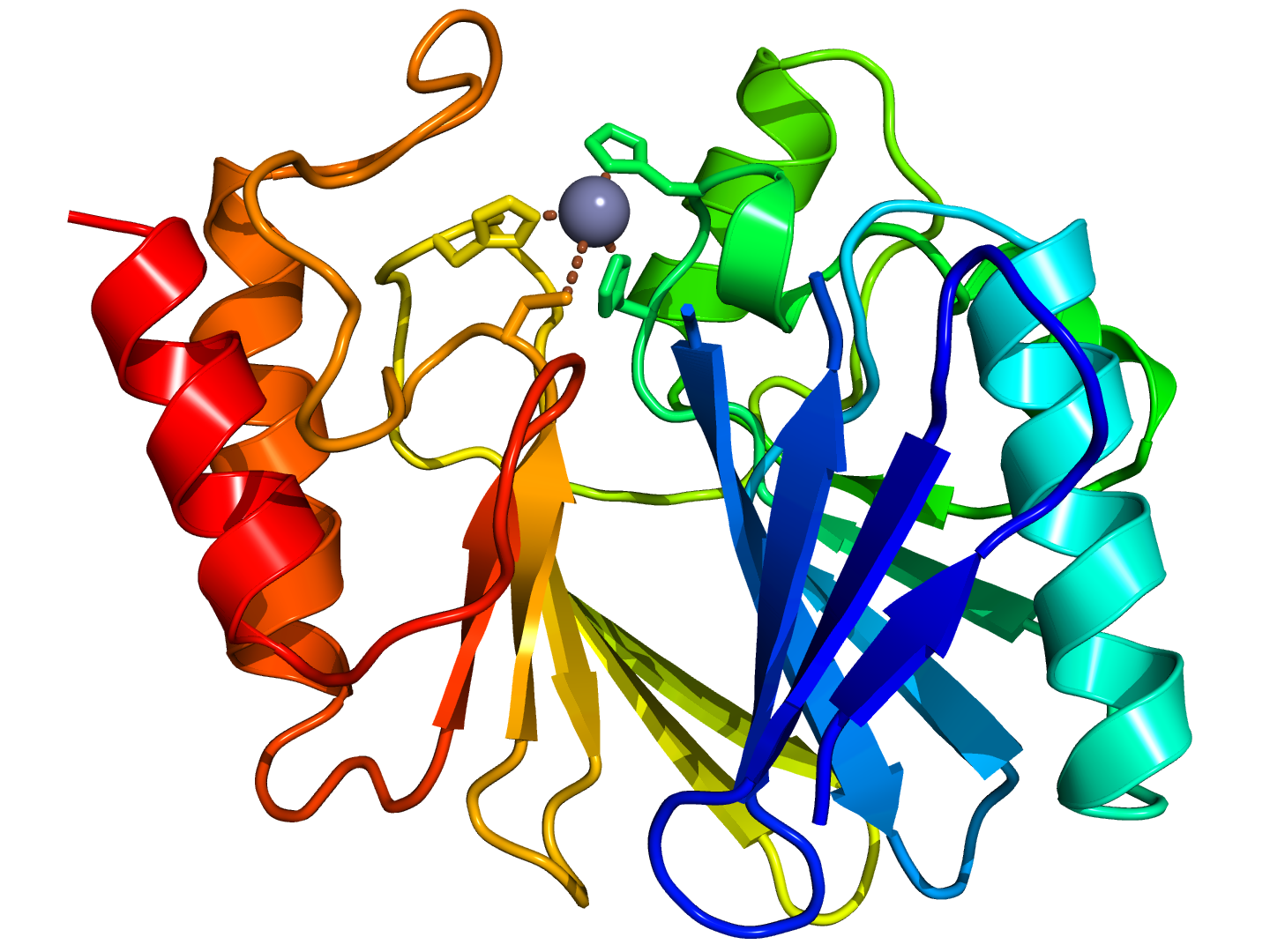
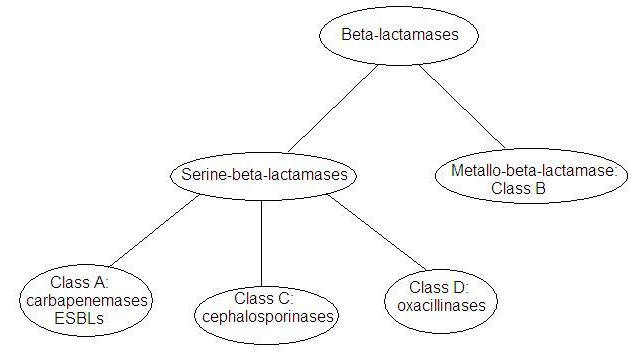
Development of metallo-beta-lactamase inhibitors - Uppsala Antibiotic Center - Uppsala University, Sweden
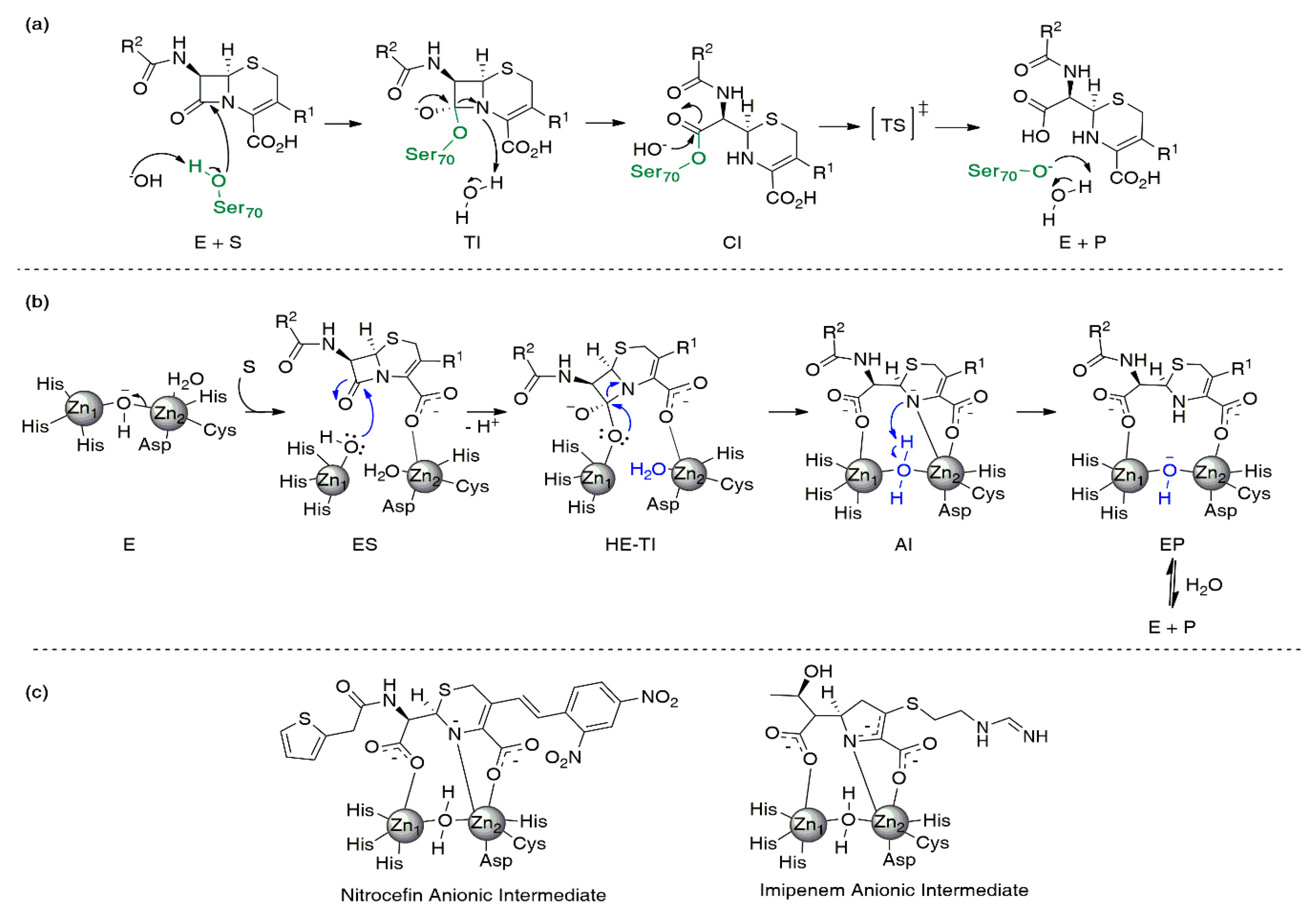
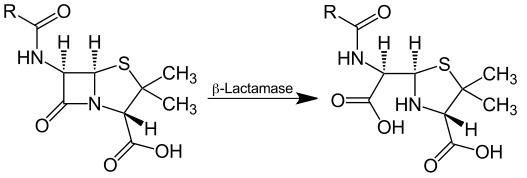
Biomolecules | Free Full-Text | Metallo-β-Lactamase Inhibitors Inspired on Snapshots from the Catalytic Mechanism
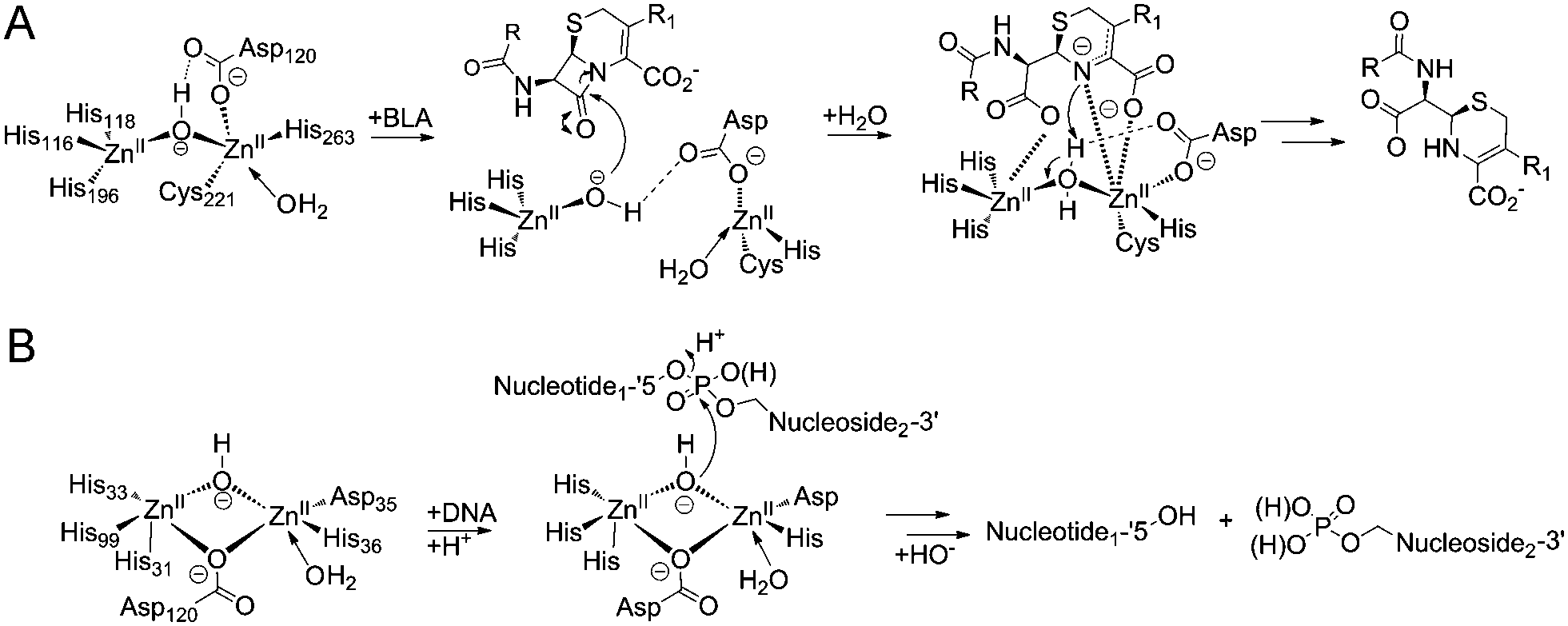
Cephalosporins inhibit human metallo β-lactamase fold DNA repair nucleases SNM1A and SNM1B/apollo - Chemical Communications (RSC Publishing) DOI:10.1039/C6CC00529B

Cross-class metallo-β-lactamase inhibition by bisthiazolidines reveals multiple binding modes | PNAS

Imitation of β-lactam binding enables broad-spectrum metallo-β-lactamase inhibitors | Nature Chemistry
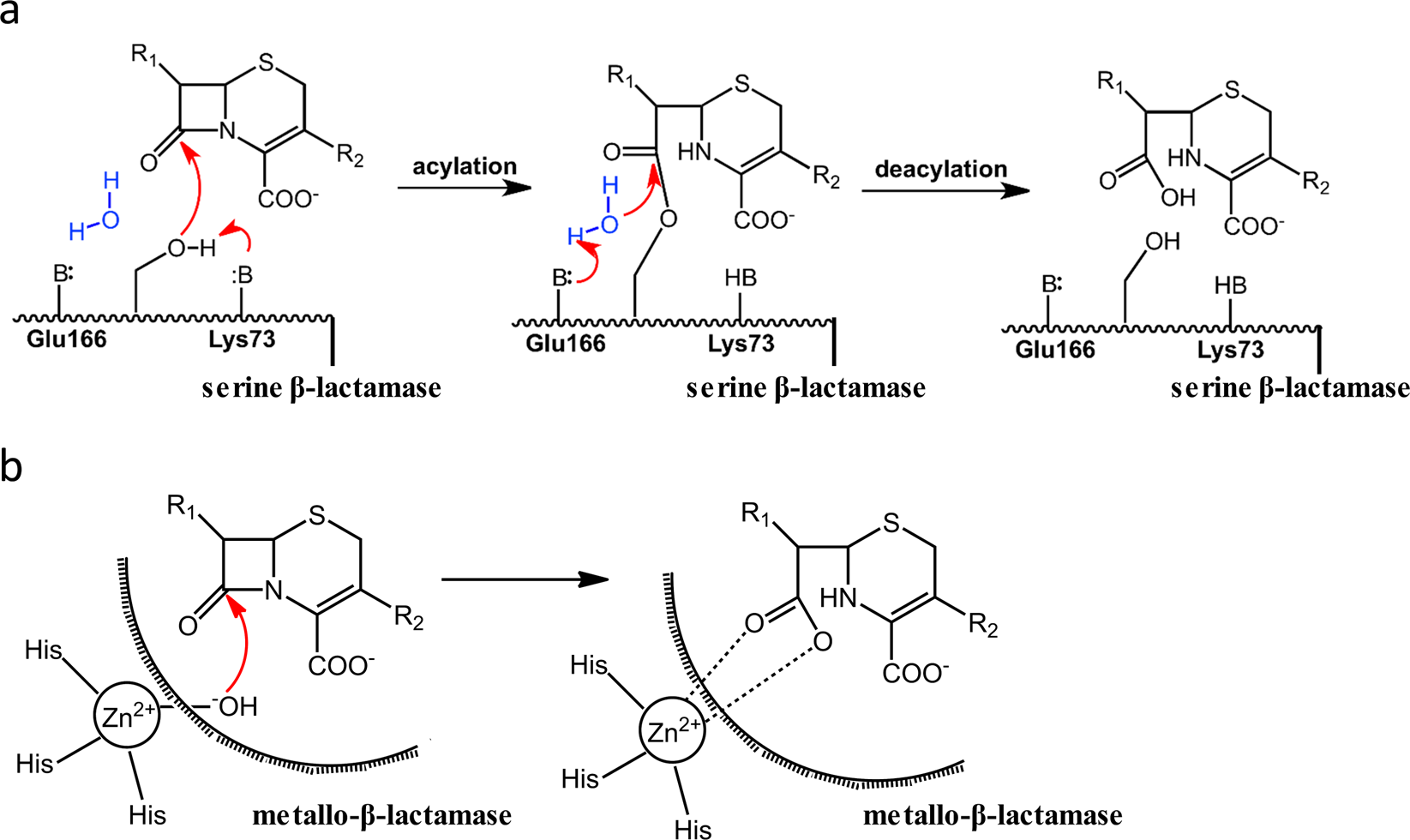
The hydrolytic water molecule of Class A β-lactamase relies on the acyl-enzyme intermediate ES* for proper coordination and catalysis | Scientific Reports

2-Mercaptomethyl Thiazolidines (MMTZs) Inhibit All Metallo-β-Lactamase Classes by Maintaining a Conserved Binding Mode | ACS Infectious Diseases

New Delhi metallo-β-lactamase-1: structure, inhibitors and detection of producers | Future Medicinal Chemistry

Figure 1 from Metallo-beta-lactamases of Pseudomonas aeruginosa--a novel mechanism resistance to beta-lactam antibiotics. | Semantic Scholar

The Continuing Challenge of Metallo-β-Lactamase Inhibition: Mechanism Matters: Trends in Pharmacological Sciences
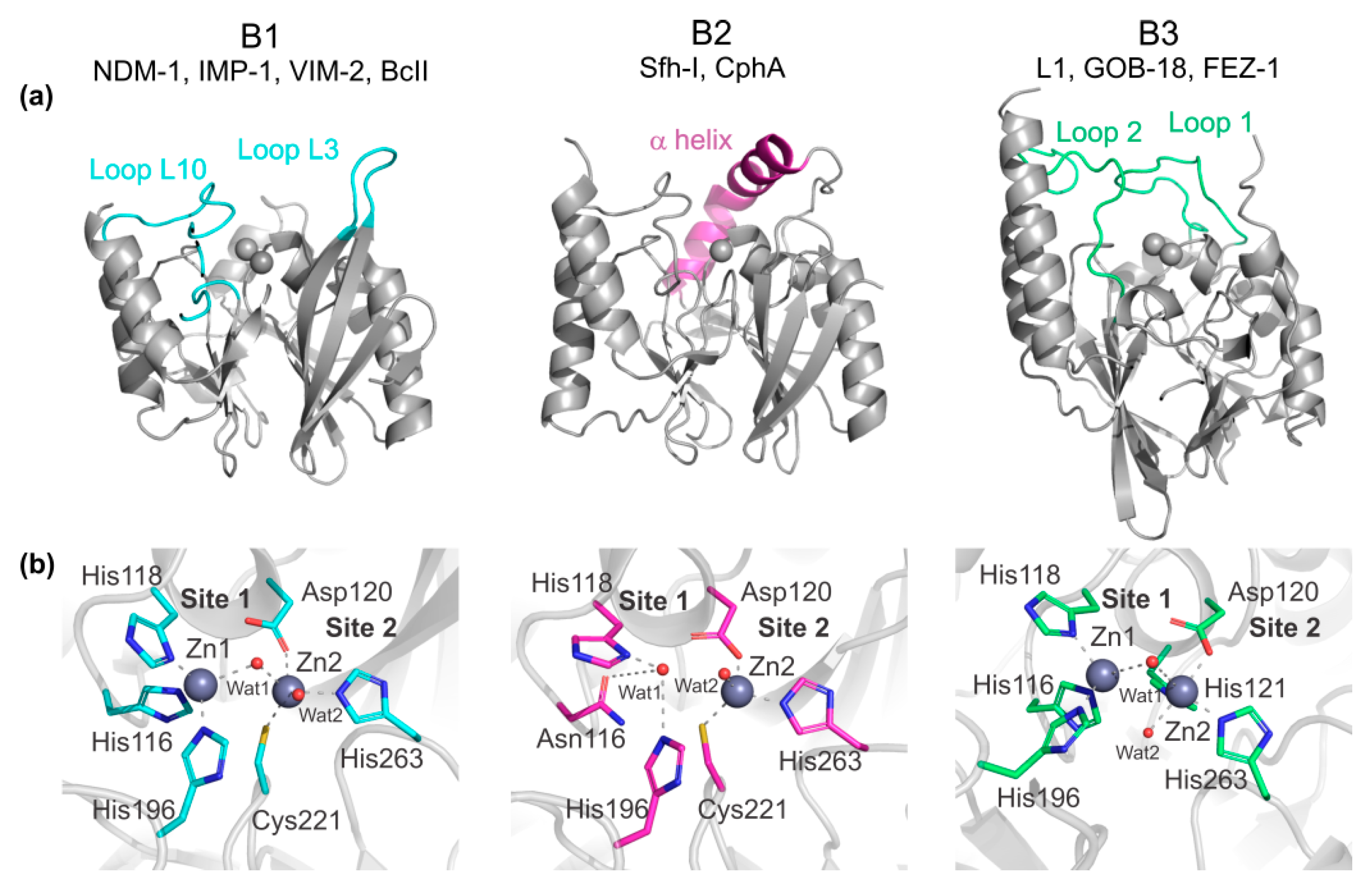
Biomolecules | Free Full-Text | Metallo-β-Lactamase Inhibitors Inspired on Snapshots from the Catalytic Mechanism